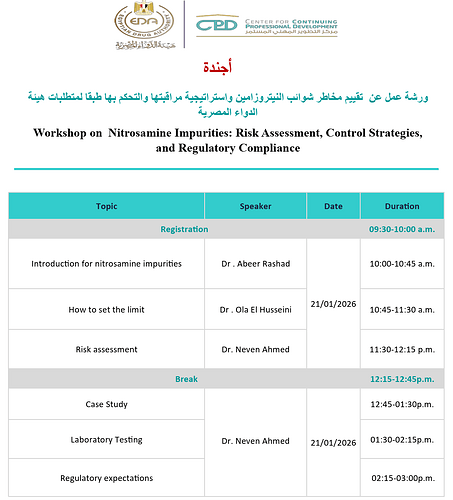
The Egyptian Drug Authority announced the opening of registration for the Workshop on Nitrosamine Impurities: Risk Assessment, Control Strategies, and Regulatory Compliance.
This workshop aims to introduce the Egyptian Drug Authority’s requirements for controlling nitrosamine impurities, focusing on their formation in raw materials and pharmaceutical products, risk assessment, and strategies to minimize them.
When: The workshop is scheduled to take place on Wednesday, January 21, 2026
Where: Egyptian Drug Authority Premises El Mansouria.
Who: The workshop is intended for companies’ representatives.
The workshop will run from 9:30 AM to 3:00 PM
The deadline for registration on Monday, January 19, 2026.
To register, please click on the following link:
Registration
The trainee will receive a certificate of attendance accredited by the Egyptian Drug Authority Center for Continuing Professional Development.