Dear Community, @Nitrosamines_Analyzer @Nitrosamines_Explorer @Nitrosamines_Investigator @Nitrosamines_Mitigator @Nitrosamines_Pioneer
I want to bring something closer to this community—because it’s the kind of topic that can’t live only inside formal notices and technical circles.
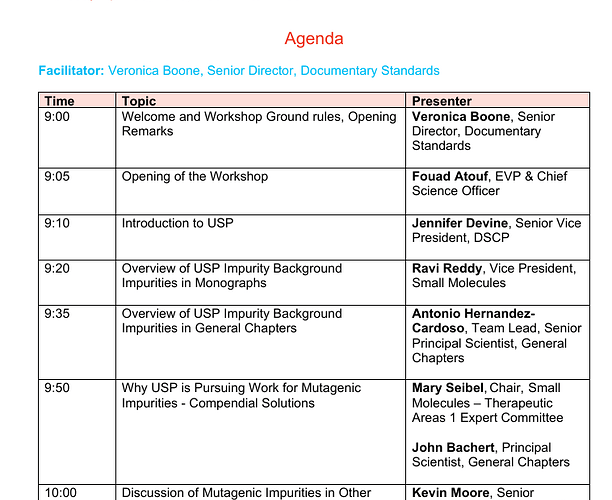
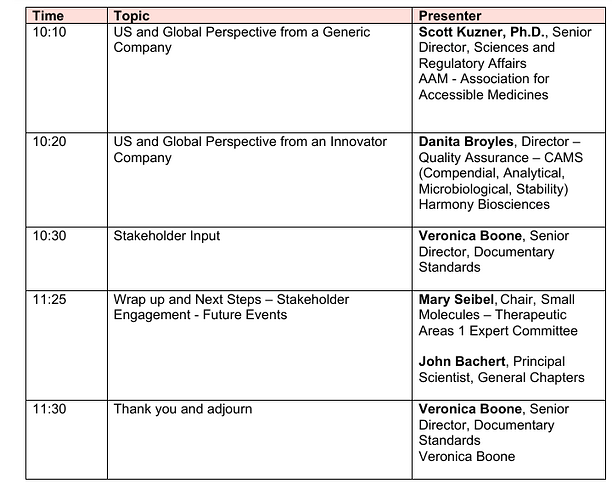
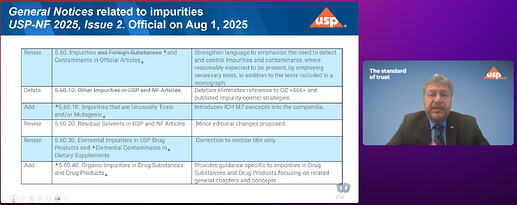
USP published last year a compendial notice announcing ongoing work to address mutagenic impurities and potentially mutagenic impurities in the USP–NF (USP Compendial notice) . You’ve worked through nitrosamines with us, you already know the feeling: this is one of those moments where the science is moving fast, the risk is real, and the compendial space has to catch up in a thoughtful way.
Here’s the honest truth: this isn’t a “should we do it?” conversation. We’re going to do it.
Because the quality and safety risk is already sitting in the room with us—sometimes quietly, sometimes loudly—across many materials and products in the supply chain.
What we’re really trying to tackle first is the harder question: how do we do it well?
And by “well,” I mean:
- in a way that actually protects patients,
- in a way that makes sense across thousands of compendial articles,
- in a way that doesn’t create confusion or unintended consequences,
- and in a way that is transparent and fair to the people who have to implement it.
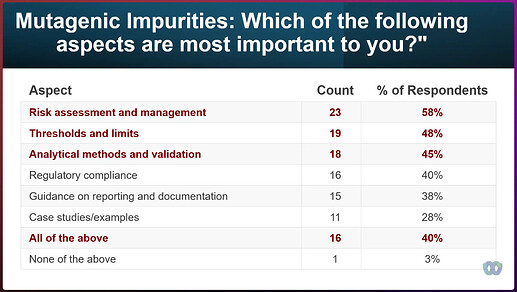
If you’ve ever tried to explain mutagenic impurities to someone outside of this niche, you know how quickly the conversation can become overwhelming, hard to explain, and, as a consequence, overlooked. We need better ways to identify, assess, and control risk—consistently and with the appropriate level of urgency. Nitrosamines has proves, this is not just about having a checklist, a limit or a testing method to follow.
USP isn’t starting from zero here. A while back, USP published a Stimuli article that laid out the challenge and invited early input (USP-PF Stimuli Article free-link)
And if you read it, you’ll see why this is not straightforward: compendial standards are public standards. They have to work globally, across different processes, different manufacturing strategies, different risk profiles, and different regulatory expectations. That complexity is real—and we’re not pretending otherwise.
Also important: We’re not walking away from ICH M7. Should I repeat that? We’re not walking away from ICH M7.
Actually, we’re doing the opposite: we’re trying to build bridges to M7 comprehensively—so the compendium can better reflect the same risk-based thinking that many of you already use every day.
Now, here’s the part I personally find most encouraging.
I’ve been at USP long enough (15 years, 2 months, and 15 days -but who is counting) to know how rigorous and structured compendial work normally is. But this effort feels different in a good way. There’s a real openness in the team—an interest in doing this with fresh eyes, and a willingness to hear perspectives that don’t always show up in traditional comment cycles.
My colleague John Bachert is leading this work, and we’re supporting him and the USP experts volunteer group that has formed around it. They’re not looking for a quick fix or a narrow solution. They’re trying to design a path that is comprehensive, transparent, and inclusive.
And that’s where I want to pull you in.
Because, just like nitrosamines, this cannot be a conversation limited to regulatory and compendial affairs teams. We need the voices of:
- analytical scientists who know what is feasible in practice,
- process chemists who understand where impurities really come from,
- toxicologists who can help keep the risk conversation grounded,
- quality leaders who have to translate guidance into systems,
- manufacturers who will have to implement this across portfolios,
- and regulators who see the real-world outcomes.
And yes—we need the stories too. The lessons learned. The messy realities. The “this looked good on paper but failed in implementation” moments.
We want to learn from the good, the bad, and the ugly—because that’s what it takes to build better public standards.
USP shared the announcement publicly today on LinkedIn (USP LinkedIn post), but I wanted to bring it here, to a community that has already proven it can handle complex impurity challenges with honesty and shared learning.
So consider this an invitation:
If you’ve been through MI/PMI strategy conversations in your company—if you’ve faced roadblocks, grey areas, disagreements, or strong lessons learned—we want to hear from you.
More soon. And thank you, as always, for being the kind of community that helps turn difficult problems into shared progress.
— Naiffer