https://jpharmsci.org/article/S0022-3549(26)00063-8/abstract
Abstract
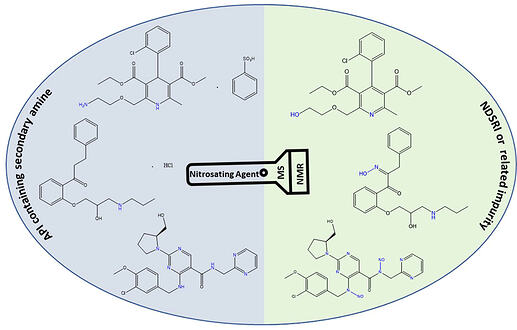
The accurate detection and quantification of nitrosamine drug substance related impurities (NDSRIs) requires validated analytical techniques. However, the availability of standards for validation is still evolving due to the structural complexity of these impurities. Thus, standards often need to be synthesized in-house and characterized to confirm identity and purity. In some cases, the synthesis of NDSRIs may follow alternative reaction pathways leading to the formation of compounds other than N-nitrosated derivatives, potentially resulting in the generation of new impurities. Among various analytical techniques, liquid chromatography-mass spectrometry (LC-MS) and nuclear magnetic resonance (NMR) spectroscopy stand out for characterization of NDSRIs due to their ability to provide detailed structural information and distinguish between different isomers. In this study, the application of LC-MS and NMR spectroscopy in the characterization of NDSRIs was explored and the identification of three new process related impurities formed during the synthesis of NDSRIs are discussed. Structural differentiation of the C- and N- nitroso analogs is difficult solely using MS techniques due to the identical molecular mass, thus the significance of employing ¹⁵N two-dimensional NMR spectroscopy for investigating impurity formation during structural characterization is emphasized here. This study shows that advanced analytical techniques help achieve more accurate identification of NDSRIs, which in turn ensures that pharmaceutical products meet high quality standards.