Sanofi decided to stop its worldwide production of Antabuse. According to the articles, Antabuse is said to contain high concentrations of the carcinogenic substance nitrosamine.
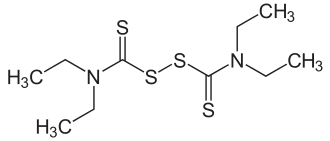
Disulfiram
Sanofi decided to stop its worldwide production of Antabuse. According to the articles, Antabuse is said to contain high concentrations of the carcinogenic substance nitrosamine.

Disulfiram
how interesting!
It is similar to the case of Chantix.
I am even more surprised because this substance had been studied as an inhibitor of certain nitrosamines…
At times it is difficult to keep up with every product that has been removed from the market and their structures.
Is this the first tertiary amine?
The potential for formation of nitrosamines with this active appears to have been identified all the way back to the early 1980s.
I would say is the 2nd case, Orphenadrine citrate was the first that I know of. However, interesting to see this cases. Normally, the “general” risk is lower than a secondary amine. But should not be completelly discarded, it is a case by case.
As the different name of Disulfiram is Bis(diethylthiocarbamoyl) Disulfide, Disulfiram may not be a simple tertiary amine but thiocarbamoyl.
And the risk assessment report by EMA mentioned NDEA contained in Disulfiram.
The MDD of Disulfiram is 500mg and 94ppb is equivarent to 47ng.
It is indeed not the “conventional” tertiary amine. Thanks for sharing.
Is Sanofi sharing the information which Nitrosamine it is? Is it NDEA?
If it is NDEA I wonder from where it is more likely to originate. From the Synthesis itself or contaminated starting material or can it be formed within the drug product during manufacturing or storage? Or in worst case, both?
For it to be withdrawn, I would bet on a problem of degradation, releasing DEA which would lead to unacceptable amounts of NDEA
I would also expect the decision to be on the commercial side. The amount of work required to put in place mitigation actions would not be worthwhile taking into account patents already (less revenue) expired and supply contrains as indicated in the press release. And I would also suppose alternatives are in the market, etc.