Dear nitrosamines exchange community,
I am currently conducting a risk assessment for trospium chloride film-coated tablets and have a few questions that you may be able to help me with.
I do not consider trospium chloride itself to be a risk because it is a quaternary ammonium compound and would first have to be dealkylated.
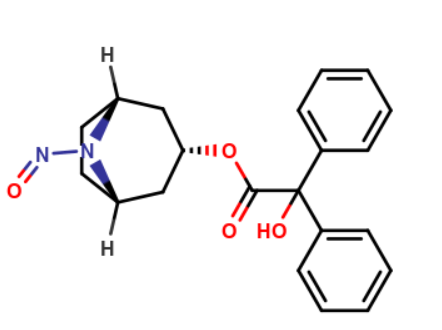
I see the starting material for trospium chloride synthesis—nortropine—and the related impurity B (benzyl acid nortropine ester) as more problematic. Both of these substances are secondary amines. Benzyl acid nortropine ester is actually always < 100 ppm, and I don’t have any analysis values for nortropine itself. However, I actually assume that it is completely converted during API synthesis and therefore does not pose a risk. When I categorize the NDSRI of benzyl acid nortropine ester (see picture below) using the CPCA approach, I end up in potency category 5 because there is only 1 alpha-hydrogen on each side of the N-nitroso group. Have I assessed this correctly? How do you view the general risk posed by this nitrosamine contamination? Based on various publications (Moser et al., 2023 – N-Nitrosamine Formation in Pharmaceutical Solid Drug Products: Experimental Observations; Carloni et al., 2023 – Solid State Kinetics of Nitrosation Using Native Sources of Nitrite) on nitrosamine formation in the solid state, a low single-digit conversion rate is to be expected.