This application note was created to aid in risk assessment and the development of control strategies for evaluating nitrite levels in excipients. It also serves as an analytical resource for regulators and the pharmaceutical industry to determine nitrite and nitrate levels in excipients and to study lot-to-lot variability
This analytical note describes a selective and sensitive ion chromatography procedure for determining nitrite and nitrate levels in Crospovidone. The analytical procedure utilizes anion exchange separation coupled with conductivity detection
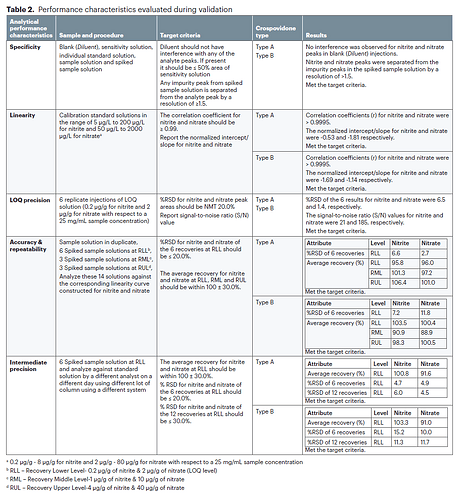
- Limit of quantitation (LOQ): 5 μg/L of nitrite and 50 μg/L of nitrate (0.2 μg/g for nitrite and 2 μg/g for nitrate with respect to a 25-mg/mL sample concentration)
- Validated range: 0.2–8 μg/g for nitrite and 2–80 μg/g for nitrate with respect to a 25-mg/mL sample concentration
- Precision: %Relative standard deviation (RSD) of nitrite and nitrate of the 6 recoveries at LOQ levels were less than 20.0%
- Accuracy: %Recovery for nitrite at 0.2, 1, and 4 μg/g and nitrate at 2, 10, and 40 μg/g were within 100 ± 30.0%
Method Performance Characteristics evaluated during validation
Application Note - Quanitification of nitrite and nitrate in Crospovidone.pdf (2.3 MB)
Disclaimer
This application note was developed to provide an analytical resource for regulators and industry to monitor nitrosamine impurities in drug products, drug substances or other matrices. It is intended to serve as a resource for informational purposes only and not as an USP-NF compendial documentary standard. This document was developed by USP staff without a public comment period and does not reflect USP or USP’s Expert Body opinions on future revisions to official text of the USP-NF. Parties relying on the information in this document bear independent responsibility for awareness of, and compliance with, any applicable federal, state, or local laws and requirements.
This is USP’s first non-compendial method published in Analytical hub for determination of Nitrite and Nitrate in Lactose. We will be posting more such methods for several at-risk excipients. This method can be used to establish level of nitrites/ nitrates in excipients and study lot to lot variability. It is intended to serve as a resource for informational purposes only and not as an USP-NF compendial documentary standard. Users will have to verify suitability of this procedure for intended purpose