As we step into this new year, I’ve found myself captivated by a scientific article that goes beyond the expected scientific discourse and ends with a deep reflection—a conclusion that I believe is worth sharing.
![]() This article revolves around medicines and the prevention of contaminations, shedding light on the tightly regulated nature of pharmaceutical quality, guided by international directives such as ICH Q1-Q14.
This article revolves around medicines and the prevention of contaminations, shedding light on the tightly regulated nature of pharmaceutical quality, guided by international directives such as ICH Q1-Q14.
These directives encompass a wide range of quality elements, spanning stability, validation, impurities, specifications, and pharmaceutical quality systems. Within this rigorously regulated framework, regulatory bodies play a crucial role in maintaining standards. However, despite these regulations, recalls of medications persist, serving as crucial protective measures for patient well-being.
The causes for these recalls often stem from breaches in Good Manufacturing Practices (GMP), microbial contamination in non-sterile products, issues with dissolution tests, or the presence of degradation products and impurities.
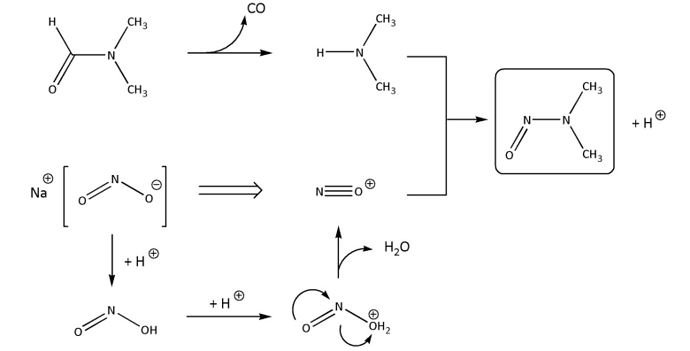
The authors meticulously detail historical instances, placing particular emphasis on nitrosamines.
What distinguishes nitrosamines is their accidental discovery in valsartan, highlighting a stark contrast between drug and food chemistry testing protocols. While nitrosamines are commonly found in food sources, their unexpected presence in medication exposed a notable gap in routine drug testing methods. Tackling these challenges necessitates advanced analytical techniques and innovative solutions.
Methods like UPLC-HRMS (QTOF) are highlighted as crucial tools for detecting unforeseen impurities, transforming the approach to testing substances in line with international pharmacopoeias.
Strategies for prevention and control pivot on risk assessments and vigilant monitoring of solvent and reagent recycling to prevent cross-contamination. Additionally, the significance of primary packaging cannot be overstated, ensuring both safety and the preservation of drug composition.
In conclusion, the pursuit of pharmaceutical quality remains a constantly evolving field, demanding continuous vigilance and innovation to guarantee patient safety. The collaboration between industry stakeholders, regulators, and scientists emerges as pivotal in anticipating and resolving quality issues.
![]() The final message of the article highlights the importance of proactive strategies, encouraging us to step up and prevent drug and product contaminations in advance. As we journey through this new year, let’s welcome the lessons learned from this thoughtful conclusion. May it motivate us to cultivate innovation, anticipate challenges, and enhance our methods for protecting the purity of medicines, benefiting everyone’s well-being.
The final message of the article highlights the importance of proactive strategies, encouraging us to step up and prevent drug and product contaminations in advance. As we journey through this new year, let’s welcome the lessons learned from this thoughtful conclusion. May it motivate us to cultivate innovation, anticipate challenges, and enhance our methods for protecting the purity of medicines, benefiting everyone’s well-being. ![]()