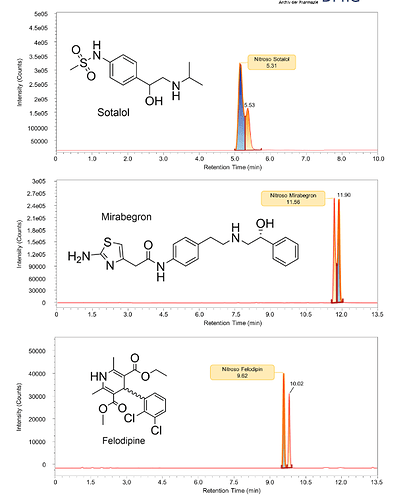
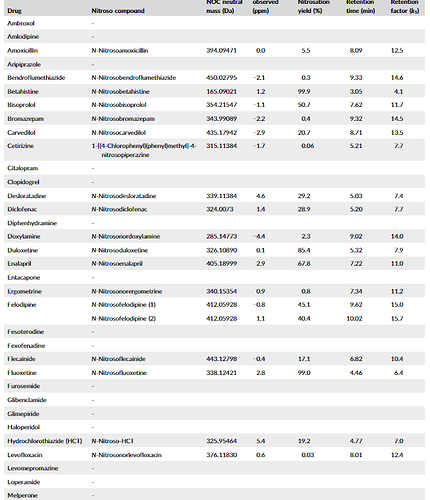
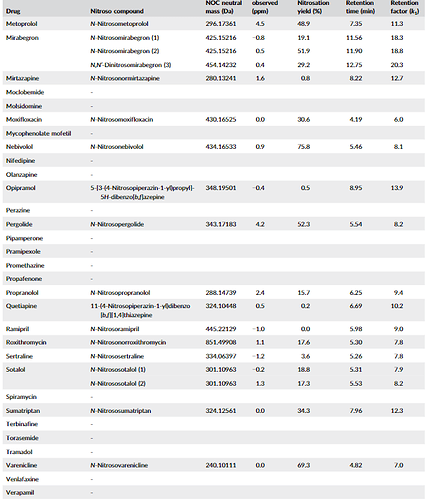
A great article by Schmidtsdorff et all. - The study incorporated a nitrosation assay procedure into high‐resolution supercritical fluid chromatography (SFC)–mass spectrometry screening to test
the potential of direct nitrosation of active pharmaceutical ingredients (APIs)
Link to access: https://onlinelibrary.wiley.com/doi/full/10.1002/ardp.202100435