The revision of 15-Sep-2022 included the following changes:
- Part B: Incorporation of DMF holder’s agreement to information exchange re. risk assessments for nitrosamine impurities as section 4.2;
- Subsections 4.1 and 4.2 for clearer structure
IDRAC_287732_20-Sep-2022_Swissmedic Form_ DMF HMV4 (ZL000_00_035_FO), 15-Sep-2022.pdf (604.0 KB)
4 Likes
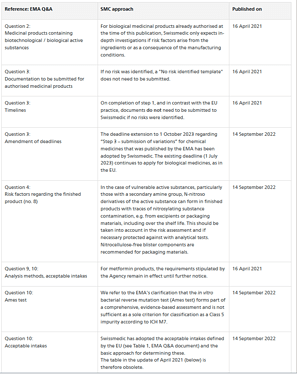
Swissmedic updated nitrosamine risk assessment policies to harmonize with those of EMA. The deadline for step3 is also delayed.
Specific requirements of Swiss healthcare may entail deviations from the EMA’s approach. Such deviations or clarifications Swissmedic deems necessary are published in the following table and will be updated on an ongoing basis.
1 Like