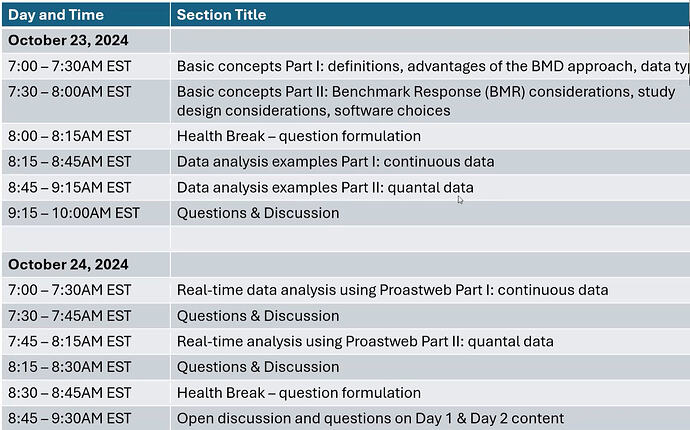
Highlighted in the potential revision of ICH M7, it’s essentail we understand (specially us not toxicologist!) what ‘Bench Mark Dose’ is.
I am sharing a recorded training I came across recently, hosted by HESI back in 2024. It features:
Paul White, Health Canada, Ottawa, Canada
Guangchao Chen, RIVM, Bilthoven, The Netherlands
George Johnson, Swansea University. Swansea, United Kingdom
John Wills, GlaxoSmithKline, Stevenage, United Kingdom
Andreas Zeller, Hoffmann-La Roche, Basel, Switzerland
The Benchmark Dose (BMD) approach has emerged as the most pragmatic and robust approach for the analysis and interpretation of toxicological dose-response data. It can be used to determine point-of departure values, sometimes called dose-response reference points, which can be used for comparative potency analysis, read across, and regulatory decision-making. Increasing use of the BMD approach for analysis and interpretation of genetic toxicity dose-response data has contributed to a great deal of interest in the basic concepts underlying the approach; moreover, the techniques used to determine BMD values. This short course will provide (i) an overview of the BMD approach and associated concepts, (ii) examples of analysis and interpretation of genetic toxicity dose-response data, (iii) real-time analysis of selected datasets.)