Drug substance-related impurities (NDSRIs) are a class of nitrosamines sharing structural similarity to the API, NDSRIs can be generated during manufacturing or during the shelf-life storage period of the drug product. In some cases, the root cause of NDSRI formation has been attributed to nitrite impurities present in excipients at parts-per-million amounts.
Most of the molecules having probability to formation of NDSRI in presence of nitrite based on their structure. Can anyone explain me is NDSRI evaluation is mandatory for nitrosamine risk assessment.
All potential Nitrosamines flagged during risk assessment should be assessed. Please check the post where we discussed the recent FDA Forum where NDSRIs where discussed.
It is obligatory unless (unlikely) there is existing data given that the nitrosamine under consideration is not mutagenic. If you have NDSRI the required actions are very costly and complicated because you need to synthesize it in order to have an analytical standard to determine the amount present in the drug product, You may also need enough for mutagenicity testing.
I agree with the previous posts. You need to evaluate the API as well as the API impurities, and product degradants. Based on the risk of nitrosamine formation, you may need to complete nitrosamine confirmatory testing on at minimum 6 batches of drug product (covering release through product expiration).
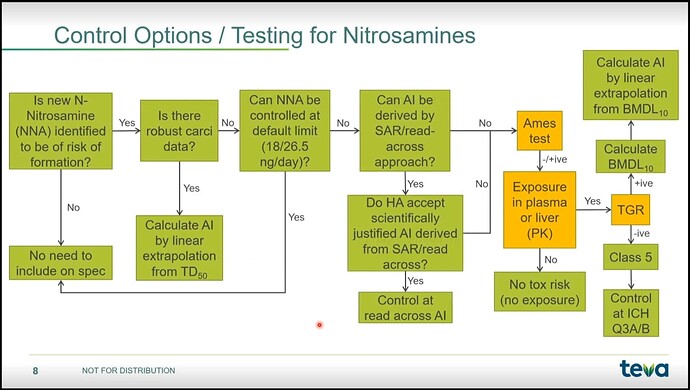
A toxicology assessment should be completed for nitrosamines not detailed in the FDA guidance to verify whether they are M7 relevant or can be covered under ICH Q3B.
In many cases you’ll need to have the nitrosamine synthesized for method validation, quantitation, and potentially Ames testing.
Hi,
In absence of interim limit by regulatory agencies (FDA, EMEA, Health Canada) for most of drug substance-related impurities (NDSRIs) ,it becomes difficult for drug product manufacture to decide on mitigation strategy (formulation or process changes).
Also what guidance should be referred for changes to mitigate nitrosamine risk (SUPAC or any other)
For example:
For one product based on API structure there is possibility of drug substance-related impurities (NDSRIs) formation. Drug product samples testing at various time period confirm presence of impurity in the range 2 to 6 ppm. Based on read-across approach (structure similarity) and mass, acceptable limit can be proposed upto 30 ppm. Confirmation for acceptance of limit of 33 ppm from agency awaited. In that case since confirmed acceptable limit not available what should be my approach.
Wait for agency reply or try to mitigate risk but upto what level
Pl suggest
@sameer for the cases without sufficient substance-specific data, agencies have established a TTC of 18 ng/day (EMA) or 26.5 ng/day (FDA) as the default option.
At the recent USP China Nitrosamine Workshop, community founder member @conudel dedicated a whole presentation to these calculations and different approaches.
Thank you very much @Naiffer_Host
Thank you so much !! It is really helpful