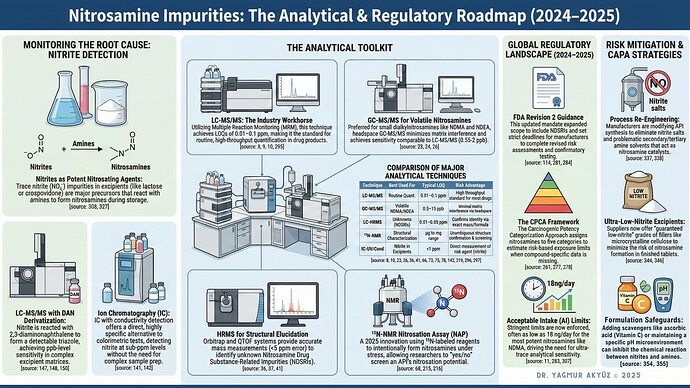
Analytical toolkit, nitrite monitoring, regulatory updates, and mitigation strategies in one visual.
A few key points:
![]() LC-MS/MS and GC-MS/MS remain our workhorses, but HRMS is becoming essential for unknown NDSRI identification
LC-MS/MS and GC-MS/MS remain our workhorses, but HRMS is becoming essential for unknown NDSRI identification
![]() Nitrite monitoring in excipients is no longer optional. It’s where many contamination stories begin
Nitrite monitoring in excipients is no longer optional. It’s where many contamination stories begin
![]() The CPCA framework and FDA Revision 2 have changed how we approach risk assessment
The CPCA framework and FDA Revision 2 have changed how we approach risk assessment
![]() ¹⁵N-NMR Nitrosation Assay (NAP) is a 2025 innovation worth watching
¹⁵N-NMR Nitrosation Assay (NAP) is a 2025 innovation worth watching
This roadmap is intended to provide perspective and support structured thinking.
Hope it’s useful for the community.