Recently, while browsing through an impurity supplier’s standards on a manufacturer’s website, I came across a particular impurity. The structure is provided below. I am wondering about the feasibility of this NDSRI in drug products. Since the CPCA isn’t suitable in this scenario, could you please advise on the AI value based on read-across?
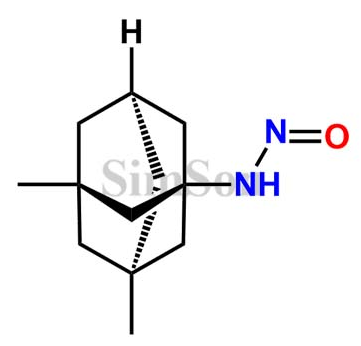 Impurity
Impurity
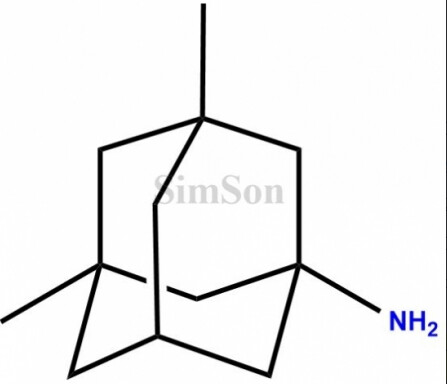
API:
Dear @Sarada.jena,
I could not find nitrosamines from primary amines in the Lhasa Carcinogenicity Data Base. I think finding an appropriate surrogate to read across isn’t easy.
I am not sure about the formation of nitrosamines from primary amines, as it is considered unstable.
I looked into this and got to the same conclusions as @Yosukemino.
A word of caution about relying on the suppliers’ list of ‘products available’ to assess potential Nitrosamines Impurities… we have got SO many reports in the community about offering structures that cannot possibly be synthesized, are not stable, or worse, the material is not what is advertised.
I’ve been working on this basis - if this changes, and we need to consider primary amines, well…
Adding the link as I forgot on first posting…
Just to mention that there was already a discussion on the site on the topic
@mflorea Thanks for remember that post from way back…
@jxl, you were the original author of the post. Do you know if the organization reported additional findings from their investigation? Any further insight will be very helpful for the folks interested in this case. Thanks.
Thank you so much for the guidance. @Yosukemino @Naiffer_Host @MarkS @mflorea
I checked with the manufacturer, their response is since there was no guidance at that moment (Dec. 2021) to address this NDSRI, they set the AI limit as 1.5ug/day.
Just a little comment based on personal experience.
Some of these suppliers which offer via website numerous N-nitrosamine impurities are, actually, synthesis on demand companies.
So, these companies tend to post in the website many API-derived N-nitrosamines. Then, when one ask for more information such as price or lead time, sometimes it comes up that they are actually not able to prepare such N-nitrosamine impurities that they offer!
Very true @Daniel-Bafaluy . Thank you very much.
Hi, @Yosukemino,
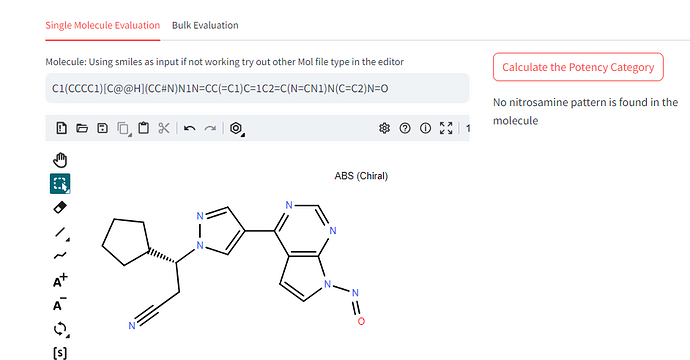
Whether N-Nitroso Ruxolitinib is possible? Is there nitrosamine pattern is found in the molecule? If Possible could you please advice, what will be the AI limit of this molecule?
Refer smiles input for structure:
C1(CCCC1)C@@HN1N=CC(=C1)C=1C2=C(N=CN1)N(C=C2)N=O
@pranavgulhane, I suggest using one of the many tools we have referenced in the community to run the calculation; you will get a lot of value from doing it. I know it is always good to validate the calculation, but doing it will benefit your understanding tremendously
Dear Naiffeer, thank you but After using calculations, I am not getting even pattern, Kindly advise. How can i consider is this nitrosamine impurity?
refer
As stated in the guidelines, the potency categorization approach does not apply to N-nitrosamines where the N-nitroso group is within an aromatic ring (for example, nitrosated indole). This is why you don’t get a result using this software.
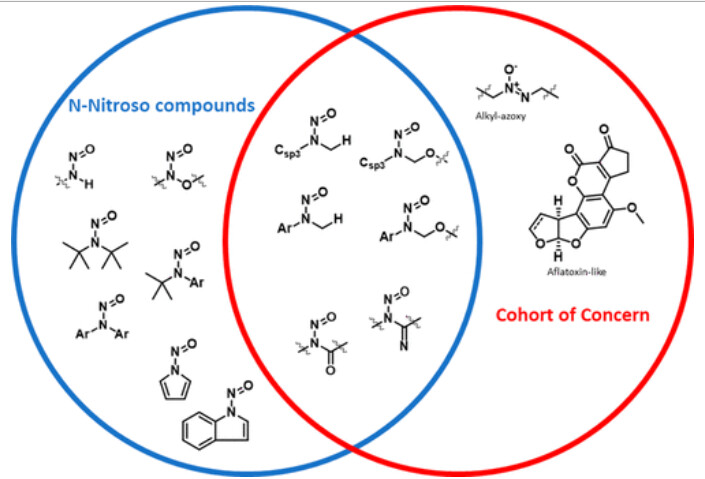
On the other hand, this kind nitroso derivative is not considered to belong to the cohort of concern, mentioned in ICH M7. Therefore, it will be controlled at TTC limit of 1.5µg/day. Check for example “Drawing a Line - Where Might the Cohort of Concern End” (Ponting & Foster, OPRD, 2023) and references cited. The diagram below, part of this article, is illustrative.
Thank you sir for demonstration.
Nevertheless, should we perform this impurity testing for the finished product and drug substance as a precautionary measure or can we abandon the study absent use in risk assessment?
If you decide to take the testing route, be extremely vigilant on where you get the reference material. The chemistry of that impurity seems tricky.
The conclusions of the risk assessment will inform if testing is required.